Alumina vs Aluminum: What’s the Difference in Properties, Composition, and Uses?
Across different regions, people spell the same metal in two ways — aluminum and aluminium. In most cases, this is only a regional language difference. However, when the discussion changes from aluminum vs aluminium to alumina vs aluminum, the topic is no longer about spelling. It becomes a comparison between two very different materials with very different structures, properties, and industrial uses.
This distinction matters in manufacturing, engineering, raw material sourcing, technical ceramics, refractories, polishing, fillers, catalysts, and many other industrial fields. Buyers often see the words aluminum, aluminium, alumina, and aluminum oxide used in different contexts. Without a clear explanation, it is easy to confuse the metal with the oxide ceramic.
This guide is written for engineers, purchasing managers, product developers, wholesalers, factories, and industrial users who need a practical understanding of how these materials differ. It explains the relationship between aluminum, aluminium, and alumina, and shows why alumina cannot be treated as simply another form of metal aluminum. Instead, it is a high-performance oxide ceramic material with its own physical behavior, chemical stability, and application value.
Table of Contents
- 1. Aluminum vs Aluminium: Why Two Spellings Exist
- 2. Quick Summary: Alumina vs Aluminum
- 3. Chemical Identity: What Each Material Really Is
- 4. Physical Properties Compared
- 5. Chemical Behavior and Stability
- 6. How Aluminum and Alumina Are Produced
- 7. Industrial Applications: Metal vs Ceramic
- 8. Why Alumina and Aluminum Cannot Replace Each Other
- 9. Comparison Table: Alumina vs Aluminum
- 10. FAQ
- 11. About BanlanChem
1. Aluminum vs Aluminium: Why Two Spellings Exist
Before comparing alumina and aluminum, it helps to clear up one common source of confusion: aluminum and aluminium are the same metal.
The two words refer to the same chemical element, with the symbol Al. The difference is mainly regional:
- Aluminum is the standard spelling in American English.
- Aluminium is the standard spelling in British English and in many international markets.
In technical documents, catalogs, safety data sheets, industrial specifications, and trade communication, both spellings are widely used. A supplier in the United States may write aluminum powder, while a supplier in Europe or the Middle East may write aluminium powder. In both cases, the meaning is the same: the metallic element Al.
The important point is this:
- aluminum / aluminium = metal
- alumina = oxide ceramic
That is why the phrase alumina vs aluminum describes a real material comparison, while aluminum vs aluminium only describes a language difference.
2. Quick Summary: Alumina vs Aluminum
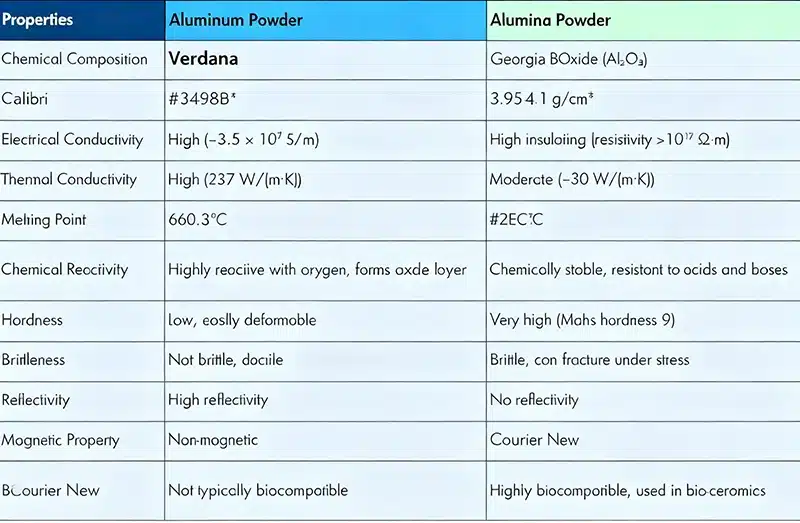
If you only remember one section of this article, remember this comparison:
- Aluminum / Aluminium (Al) is a metal. It is lightweight, conductive, reflective, and easy to shape.
- Alumina (Al2O3) is a ceramic oxide. It is hard, heat-resistant, chemically stable, wear-resistant, and electrically insulating.
Although alumina is made from aluminum-containing raw materials, it is no longer a metal. Once aluminum is chemically combined with oxygen to form aluminum oxide, the material category changes completely. The resulting oxide has different bonding, different structure, different performance, and different uses.
This is why aluminum is commonly used in structural parts, packaging, transportation, electrical systems, and consumer products. Alumina, by contrast, is commonly used in abrasives, polishing materials, refractories, technical ceramics, catalyst supports, fillers, and insulating parts.
If you want a broader introduction to alumina itself, you can also read What Is Alumina Oxide? or review BanlanChem’s Alumina Powder product page.
3. Chemical Identity: What Each Material Really Is
Aluminum / Aluminium (Al)
Aluminum is a metallic element. It is part of the periodic table and exists as a metal with metallic bonding. Because of its free-moving electrons, aluminum has good electrical conductivity and thermal conductivity. It is also relatively light compared with many other engineering metals.

One important feature of aluminum is that it reacts with oxygen very quickly. When exposed to air, it forms a very thin oxide film on the surface. This oxide layer helps protect the underlying metal from further corrosion in many environments.
However, that thin surface oxide is not the same thing as bulk alumina powder used in industrial applications. The surface film is only a natural protective layer. Industrial alumina is produced intentionally and processed as its own material.
Alumina (Al2O3)
Alumina is aluminum oxide. Its chemical formula is Al2O3. It is not a metal. It is an oxide ceramic material formed by the strong chemical bonding of aluminum and oxygen atoms in a stable crystal structure.
Because of this structure, alumina behaves very differently from aluminum metal. It has:
- very low electrical conductivity
- high hardness
- high melting point
- strong wear resistance
- good chemical stability
- excellent performance at elevated temperatures
This is the key reason why alumina is used in ceramics, kiln furniture, polishing compounds, abrasives, catalyst carriers, fillers, and high-temperature industrial systems. To understand its crystal behavior in more detail, see Alumina Bonding Types: Ionic vs Covalent in Al2O3.
4. Physical Properties Compared
The difference between alumina and aluminum becomes much clearer when their physical properties are compared side by side.

Appearance
Aluminum usually appears as a silver-gray metal with a metallic shine. It can be formed into sheets, powders, rods, cast parts, or extrusions.
Alumina usually appears as a white powder, granule, or ceramic-like solid depending on grade and form. It does not have metallic luster.
Electrical Conductivity
Aluminum is a good electrical conductor. This makes it useful in electrical systems, conductive parts, and heat transfer applications.
Alumina is generally an electrical insulator. This is why it is often used in insulating components and ceramic electronic parts.
Density
Aluminum has a density of about 2.7 g/cm³, which is one reason it is considered a lightweight engineering metal.
Alumina has a density of about 3.9 g/cm³, depending on the grade and form. It is heavier than aluminum, but it is also much harder and more temperature-resistant.
Hardness
Aluminum is relatively soft compared with ceramics. It can be machined and formed easily, which is one of its industrial advantages.
Alumina is very hard. In many applications, this high hardness makes it suitable for grinding, polishing, wear-resistant parts, and abrasives. For example, products such as White Fused Alumina are widely used where hardness and cutting performance matter.
Melting Point
Aluminum melts at about 660°C.
Alumina has a melting point around 2050°C, which is one of the reasons it is widely used in refractory and high-temperature environments. For a deeper explanation, see Why Alumina Has High Melting Point?
General Property Difference
These differences explain why aluminum and alumina are used in completely different product categories. One is a structural and conductive metal. The other is a stable and wear-resistant ceramic oxide.
5. Chemical Behavior and Stability
Chemical Behavior of Aluminum
Aluminum is reactive, even though it often appears stable in normal environments. It reacts rapidly with oxygen to form a protective oxide layer. It may also react with strong acids and strong bases under certain conditions. In fine powder form, aluminum can be much more reactive than solid bulk metal.
Because of its chemical activity, aluminum powder can present combustion or ignition risks in certain industrial environments. This is especially important in powder handling, fireworks, thermite systems, pigments, and metal processing.
Chemical Behavior of Alumina
Alumina is much more stable chemically. It does not behave like metallic aluminum. It does not burn like metal powder, and it is widely used in environments where chemical resistance and thermal stability are important.
Alumina is valued because it:
- resists corrosion in many industrial systems
- maintains stability at high temperatures
- performs well in ceramic and refractory environments
- supports catalytic activity in catalyst carrier systems
- provides long-term performance in fillers and insulation uses
This stability is one of the main reasons alumina is used in advanced material systems. Its role is not based on conductivity or metal processing, but on structural stability and chemical performance. BanlanChem also offers related adsorption-grade materials such as Activated Alumina and Activated γ-Al2O3 Powder for catalyst and adsorption applications.
6. How Aluminum and Alumina Are Produced
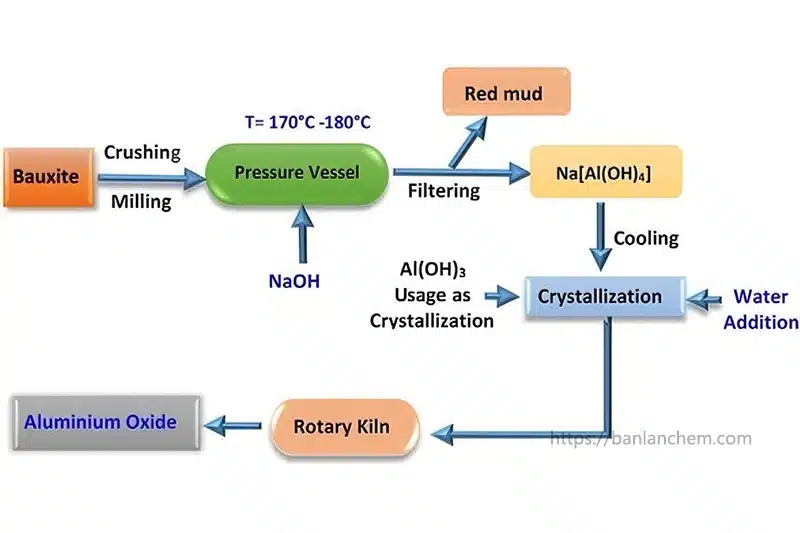
How Alumina Is Produced
Industrial alumina is commonly produced from bauxite ore through the Bayer process. In simplified terms, the process includes:
- Crushing and preparing bauxite
- Extracting aluminum hydroxide
- Purifying the material
- Calcining the hydroxide at high temperature
- Producing alumina powder
If you want a more detailed process explanation, you can also read What Is Calcined Alumina Powder and How It’s Made? and Types of Alumina for Industrial Use.
How Aluminum Is Produced
Aluminum metal is produced through a different route. After alumina is refined from ore, it can be converted into metallic aluminum through electrolytic reduction. This is a completely separate industrial step from alumina production.
Aluminum powder can then be made through methods such as atomization, milling, centrifugal processing, and other controlled powder production technologies.
This means that even though alumina and aluminum are related in the broader supply chain, they are not interchangeable materials.
7. Industrial Applications: Metal vs Ceramic
Common Uses of Aluminum
Because aluminum is conductive, lightweight, and easy to form, it is widely used in:

- transportation parts
- structural components
- electrical and thermal systems
- packaging
- consumer goods
- metal coatings
- metallic pigments
- alloy production

In powder form, aluminum may also be used in specialized pyrotechnic, coating, and metallurgical applications.
Common Uses of Alumina
Alumina is used in completely different industrial areas. Common applications include:
- abrasive materials
- polishing compounds
- refractory linings
- kiln furniture
- ceramic substrates
- insulating parts
- filler materials for plastics and rubber
- catalyst supports
- adsorbents
- filtration-related media
Many industrial buyers first encounter alumina in the form of calcined alumina, activated alumina, ATH filler, boehmite, or high purity alumina, depending on their sector. For practical reading, see What Is Alumina Powder Used For?, Aluminum Oxide Uses Explained, and How to Use Alumina Powder for Polishing.
In refractory systems, materials such as Tabular Alumina and Calcined Alumina are preferred because of their heat resistance and stability. You can also explore Tabular Alumina for Refractory Applications for a focused application guide.
8. Why Alumina and Aluminum Cannot Replace Each Other
People sometimes assume that because alumina comes from aluminum, the two materials may be used in similar ways. In real industrial practice, this is not correct.
Why aluminum cannot replace alumina
- Aluminum is too soft for abrasive uses.
- Aluminum melts too easily for refractory applications.
- Aluminum conducts electricity, so it is unsuitable where insulation is required.
- Aluminum is not stable enough for many oxide-ceramic environments.
Why alumina cannot replace aluminum
- Alumina does not provide metallic conductivity.
- Alumina cannot be used as a lightweight structural metal.
- Alumina is brittle compared with formed metal parts.
- Alumina is not suitable for applications that depend on metal ductility or shaping performance.
In short, the two materials solve different problems. Aluminum is a functional engineering metal. Alumina is a functional oxide ceramic.
This is why professional factories, suppliers, and manufacturers always separate the two categories clearly in their production systems, technical documents, and product lines.
9. Comparison Table: Alumina vs Aluminum
| Property | Aluminum | Alumina |
|---|---|---|
| Chemical formula | Al | Al2O3 |
| Material type | Metal | Oxide ceramic |
| Appearance | Silver-gray metallic | White powder or ceramic solid |
| Electrical conductivity | High | Very low / insulating |
| Thermal conductivity | High | Lower than aluminum |
| Density | About 2.7 g/cm³ | About 3.9 g/cm³ |
| Hardness | Relatively soft | Very hard |
| Melting point | About 660°C | About 2050°C |
| Chemical stability | Moderate / reactive surface behavior | High stability |
| Typical applications | Metal parts, transport, packaging, conductive systems | Ceramics, abrasives, refractories, polishing, fillers, catalyst supports |
This table shows why the phrase alumina vs aluminum is a real material comparison rather than a naming difference.
10. FAQ
Is aluminum the same as alumina?
No. Aluminum is a metal element, while alumina is aluminum oxide, a ceramic material.
Why do some people spell it aluminium?
Aluminium is the British and international spelling, while aluminum is the American spelling. Both mean the same metal.
Which is harder, aluminum or alumina?
Alumina is much harder than aluminum and is widely used in abrasive and wear-resistant applications.
Can aluminum powder ignite?
Yes, under certain conditions fine aluminum powder can be reactive and may pose ignition risks. Handling conditions matter.
Is alumina conductive like aluminum?
No. Aluminum is conductive, while alumina is generally used as an insulating material.
Is alumina safe?
Alumina is generally considered chemically stable and non-flammable in many industrial applications. Safe handling still depends on grade, particle size, dust control, and application conditions.
Where is alumina commonly used?
Alumina is widely used in ceramics, polishing, abrasives, refractories, catalyst supports, fillers, and technical industrial materials.
11. About BanlanChem
BanlanChem specializes in alumina-based materials for industrial applications. Our product range includes alumina powder, aluminum hydroxide, pseudoboehmite, boehmite alumina, activated alumina, and related material solutions for ceramics, catalysts, fillers, chemicals, and advanced applications.

We support stable supply, technical communication, and product matching for different industrial uses. If you are looking for alumina materials for polishing, refractories, fillers, catalyst systems, or specialty applications, BanlanChem can provide relevant product support and technical information.
Conclusion
The difference between alumina and aluminum is not a small technical detail. It is a major material distinction that affects performance, safety, processing, and end use.
Aluminum is a lightweight metal with conductivity, formability, and broad structural value.
Alumina is a hard, stable, heat-resistant oxide ceramic with strong value in abrasives, refractories, fillers, polishing, and catalyst-related uses.

Meanwhile, aluminum and aluminium are simply two spelling versions of the same metal. They should not be confused with alumina, which is an entirely different industrial material.
For engineers, buyers, and industrial decision-makers, understanding this difference helps prevent sourcing mistakes, improves technical selection, and makes it easier to match the right material to the right application.















